If Nature is the sculptor of life, living tissues are the best materials to sculpture – they are both pliable enough to be remodeled and yet strong enough not to be crumbled, leading to amazingly diverse life forms that are seemingly fragile yet extremely robust. This can be attributed to the ability of cells in adapting their individual or collective properties and behaviors according to the physical forces applied to them, a phenomenon happening frequently during both development and homeostasis and contributing to various human diseases when disrupted. For example, it has long been observed that lack of physical activities leads to weakening of muscles and bones, such as in the case of paralyzed patient and astronauts in the space. However, the molecular mechanisms underlying the mechanosensitive biological changes has been poorly studied.
As biological sciences become more and more intertwined with other disciplines such as physics and engineering, we are starting to appreciate that physical forces, just like biochemical signals, can activate cellular cascades and play an instructive role in regulating cell behaviors. Our long term goal is to understand how mechanical tension is sensed and translated into cellular activities, and eventually impacts the tissue morphology and behaviors. We use developing Drosophila embryos as the model system, where a lot of tissue sculpturing happens! We employ multidisciplinary approaches such as quantitative live and immunochemistry microscopy, biophysics, computational biology, molecular biology and genetics.
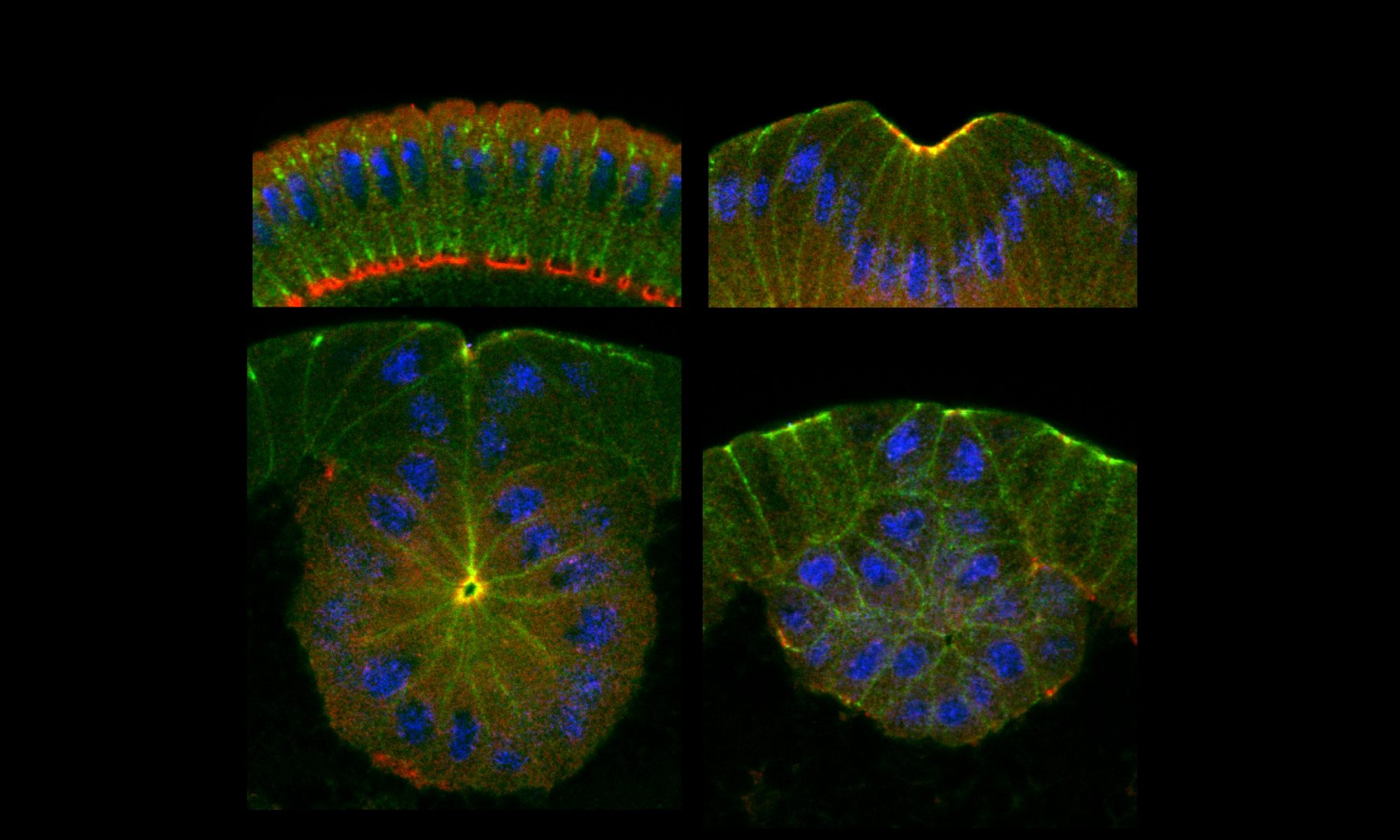
One current emphasis in the lab is to study the role of tension during two simultaneous but mechanically contradicting processes: the epithelial folding that drives the internalization of mesoderm, and epithelial-mesenchymal transition (EMT) that generate various mesodermal tissues. EMT is a process that transform closely connected epithelial cells (solid) into migratory mesenchymal cells (fluid). It is essential in generating cell diversity during both embryogenesis and evolution, but is also the major driver during cancer progression. While epithelial folding requires the strong cell-cell junctions, the final goal of EMT is to disassemble junctions. Our previous study shows the mechanosensitive remodeling of adherens junctions provides the mechanism to reconcile these two processes.